Titrations
A titration is a laboratory method in which a substance with a known concentration is added to a substance with an unknown concentration (but a known volume) until they neutralize each other. One of the substances will be an acid and one will be a base so that neutralization can occur. Titrations are done in order to obtain the concentration of the substance with the unknown concentration.
How to conduct a titration
Here is a procedure for a titration in which the substance with the unknown concentration is a base and the substance with known concentration (the one going through the burette) is an acid
First, you have to gather the necessary materials for the titration
Materials
70 mL of substance of base
100 mL of acid
2 250 mL beakers, one labeled B for the base and one labeled A for the acid
50 mL burette
Burette stand
250 mL flask
Funnel
A volume pipette that can hold 10 mL
Phenolphthalein indicator
After all the materials are gathered, put on safety goggles and tie up you hair if it is long
First, you have to gather the necessary materials for the titration
Materials
70 mL of substance of base
100 mL of acid
2 250 mL beakers, one labeled B for the base and one labeled A for the acid
50 mL burette
Burette stand
250 mL flask
Funnel
A volume pipette that can hold 10 mL
Phenolphthalein indicator
After all the materials are gathered, put on safety goggles and tie up you hair if it is long
Then, you have to clean all your materials. This is very important to the experiment, so don't skip it!
Now it's time to set up the burette for the experiment
The first titration that is done is called a rough titration. This titration is conducted in order to get the general range of how much acid must be added to base in order for neutralization to occur. Here is a step by step procedure for how to conduct a rough titration. Watch the corresponding video for each step to see a demonstration of it
1. Use the volume pipette to extract 10 mL of base from the beaker labeled "B"
- Clean both of the beakers with water, then dry them thoroughly so that neither of the substances get diluted by water left in the beakers
- Wash out the beaker with water, but you don't need to dry it
- Rinse out the burette and funnel with water to prevent contamination then with the acid in order to prevent dilution
- Rinse out the volume pipette with water then with the base
- While you're doing this, make sure your burette and volume pipette are functioning properly!
Now it's time to set up the burette for the experiment
- Put the burette into the burette stand so that the burette is high enough for the flask to fit below it
- Put the funnel into the top of the burette
- Put 70 mL of base into the beaker labeled "B"
- Put 100 mL of acid into the beaker labeled "A"
The first titration that is done is called a rough titration. This titration is conducted in order to get the general range of how much acid must be added to base in order for neutralization to occur. Here is a step by step procedure for how to conduct a rough titration. Watch the corresponding video for each step to see a demonstration of it
1. Use the volume pipette to extract 10 mL of base from the beaker labeled "B"
2. Use the volume pipette to put the 10 mbase into the flask
3. Put 3 drops of phenolphthalein indicator into the base in the flask (the substance should turn purple)
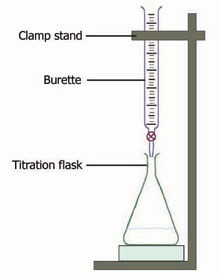
4. Put the flask with the base and the indictaor under the burette. Your set up should look something like the picture below, but with a funnel at the top of the burette
5. Pour the acid from beaker A into the burette through the funnel. Pour enough acid so that the bottom of the meniscus (see picture below) is at a line about three quarters of the way to the top of the burette. Record how much acid is in the burette at the start.
6. Turn on the burette's tap so that the acid pours in a constant stream into the base in the flask. Someone should be shaking the flask and watching its color. When they see the color start to turn from purple to clear, the burette should be turned off because the mixture is now neutral. Record how much acid is in the burette after the tap is turned off.
Now subtract the amount of acid from when after the tap was turned off from the original amount of acid to get the amount of acid used in the rough titration. This number gives us a general range for how much of the acid is needed to neutralize the base. We subtract two from the number to get the point at which we will turn the tap in real titrations so that it goes from a constant stream to a slow drip. For example, if we get 10 mL in the rough titration, we would turn the tap from a stream to a drip after 8 mL of acid have been poured from the burette into the flask. We would then turn off the tap when a color change is observed, this time getting a much more accurate reading because the acid was coming out more slowly.
Calculating concentration based on your results
Once you do a few real titrations, you can use your data calculate the average volume of acid used in the titration. This number can be used to calculate the concentration of the unknown substance using the following equation:
(Molarity of acid)*(Volume of acid)=(Molarity of base)(Volume of base)
Molarity is a measure of concentration that measures moles of a certain substance per liter and it is the measure of concentration that will be used for the answer to this equation. For the equation we're about to solve, we will say the molarity of the acid was 2 M. The volume of the base used in the procedure that was outlined was 10 mL, so that would be the volume of the base in the equation. The volume of the acid would be the average volume of acid used in the titration, which, for the purposes of solving the equation, we will say is 5 mL. So the variable in this equation would be the molarity of the base, and it would be solved for like this:
(2 M)*(5 mL)=(x)*(10 mL)
10=10x
x=1 M
And that's the concentration of our base!
(Molarity of acid)*(Volume of acid)=(Molarity of base)(Volume of base)
Molarity is a measure of concentration that measures moles of a certain substance per liter and it is the measure of concentration that will be used for the answer to this equation. For the equation we're about to solve, we will say the molarity of the acid was 2 M. The volume of the base used in the procedure that was outlined was 10 mL, so that would be the volume of the base in the equation. The volume of the acid would be the average volume of acid used in the titration, which, for the purposes of solving the equation, we will say is 5 mL. So the variable in this equation would be the molarity of the base, and it would be solved for like this:
(2 M)*(5 mL)=(x)*(10 mL)
10=10x
x=1 M
And that's the concentration of our base!