Words in blue are important terms
Basics of pH and Strength of Acids
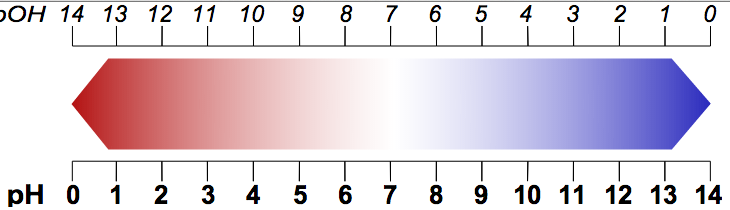
The pH scale, as shown in the picture above, is a way to indicate the STRENGTH of an acid. The strength of an acid refers to its tendency or ability to lose an H+ proton. This isa ability to lose a proton determined by the acid's ability to completely DISSOCIATE to form hydrogen ions. Dissociation is when H+ protons split away from other molecules. A strong acid will completely dissociate, while a weak acid will only partly dissociate, as demonstrate in this image
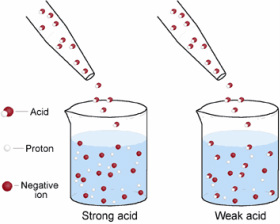
More of the molecules in the strong acid are able to completely dissociate to create H+ protons than molecules in the weak acid are (weak acid only partially dissociates)
All acids are below 7 on the pH scale. Strong acids are assigned a pH between 1 and 3, while weak acids are assigned a pH of 4 through 6.
Concentration
When an acid is able to completely dissociate, it creates more H+ protons than an acid that can only partially dissociate. This means that this acid has a higher CONCENTRATION of H+ molecules. This concentration is written as [H+] and because it is directly related to pH, it can be used to calculate pH, which you will see later.
Reducing something's concentration by adding another substance to it is called DILUTION. Usually water is used to dilute an acid or base. But be careful, because you can unwittingly dilute an acid or base and that you are using in an experiment and mess up your results if you forget to dry your materials!
Bases on the pH scale
If a substance is 7 on the pH scale, it is neutral, meaning it is neither an acid nor a base (water, for example, is neutral). When a substance is higher than a 7 on a pH scale, the substance is basic. A weak base is assigned a pH between 8 and 10, while a strong base is assigned a pH between 11 and 14.
pOH and Concentration in Bases
The pOH scale is essentially the pH scale, but backwards. This is shown in the following image:
On the pOH scale, all bases are below 7. A strong base would be between 1 and 3, while a weak base would be between 4 and 6. 7 is still neutral and above 7 is an acid. A weak acid would be between 8 and 10 and a strong acid would be between 11 and 14.
Just like for acids, the concentration of bases can is directly related to pOH and can be used to calculate it. Concentration in bases is the concentration of OH- rather than H+ and is written as [OH-].
Just like for acids, the concentration of bases can is directly related to pOH and can be used to calculate it. Concentration in bases is the concentration of OH- rather than H+ and is written as [OH-].
Calculating pH, pOH, [H+] and [OH-]
All substances have a pH number, a pOH number, an [H+] concentration, and an [OH-] concentration. Once you have one of them, you can use these equations to calculate the others.
|
Calculating pH and pOH
To calculate pH from [H+] pH= -log[H+] To calculate pOH from [OH-] pOH= -log[OH-] To calculate pOH from pH pOH=14-pH To calculate pH from pOh pH=14-pOH Sample Calculation |
Calculating [H+] and [OH-]
To calculate [H+] from pH [H+]= inverse log(-pH) which is the same as [H+]= 10^-pH To calculate [OH-] from pOH [OH-]= inverse log(-pOH) which is the same as [OH-]= 10^-pOH |
If you look at those equations and think
..then take a look at this sample calculation!
Let's say we start with [OH-]=1.29e-4
1. First, we calculate the pOH. Type the negative function into your calculator, then click the log button. Then type in the [OH-] and press enter
pOH= -log[OH-]
pOH= -log[0.000124]
pOH=3.89
2. Then, we can calculate the pH.
14-pOH=pH
pH=10.11
3. Finally, we can calculate the [H+]. Click the second function button on your scientific or graphing calculator then click the log button. Then, type in the negative sign, then the pH, and finally press enter.
[H+]=10^-pH
[H+]=10^-10.11
[H+]=7.7e-11
Now we have all of our answers
[OH-]=1.29e-4
[H+]=7.7e-11
pOH=3.89
pH=10.11
If you think you finally get all of this now, click the button below to head over to the quiz on these calculations, or if you want to keep reading about acids and bases, click next page
Let's say we start with [OH-]=1.29e-4
1. First, we calculate the pOH. Type the negative function into your calculator, then click the log button. Then type in the [OH-] and press enter
pOH= -log[OH-]
pOH= -log[0.000124]
pOH=3.89
2. Then, we can calculate the pH.
14-pOH=pH
pH=10.11
3. Finally, we can calculate the [H+]. Click the second function button on your scientific or graphing calculator then click the log button. Then, type in the negative sign, then the pH, and finally press enter.
[H+]=10^-pH
[H+]=10^-10.11
[H+]=7.7e-11
Now we have all of our answers
[OH-]=1.29e-4
[H+]=7.7e-11
pOH=3.89
pH=10.11
If you think you finally get all of this now, click the button below to head over to the quiz on these calculations, or if you want to keep reading about acids and bases, click next page