Acids and bases have three different definitions based on three different theories about them. Here are the three theories and the definitions that are based off of them.
Arrhenius Theory
The Arrhuenius theory was the first modern theory about the definitions of acids and bases that was proposed. It is not considered to be the most accurate today because it only refers to aqueous acids and bases. According to this theory,
Acid: produces hydrogen ions when put in water
Base: produces hydroxide ions when put in water
Acid: produces hydrogen ions when put in water
Base: produces hydroxide ions when put in water
Lewis Theory
In 1923, Gilbert Lewis published his definitions of acids and bases. These definitions are considered to be the most accurate definitions available today.
Acid: An electron pair acceptor
Base: An electron pair donator
Acid: An electron pair acceptor
Base: An electron pair donator
Bronsted-Lowry Theory
The Bronsted-Lowry theory of acids and bases was also proposed in 1923. According to this theory,
Acid: donator of protons (specifically positively charged hydrogen ions)
Base: acceptor of protons (specifically positively charged hydrogen ions)
Acid: donator of protons (specifically positively charged hydrogen ions)
Base: acceptor of protons (specifically positively charged hydrogen ions)
Here are the definitions of other terms that relate to acids and bases
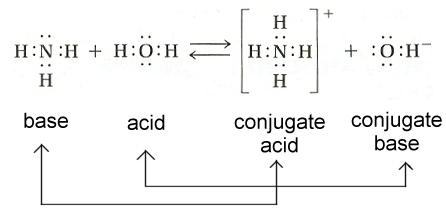
Conjugate Acids and Bases
When acids and bases react, they create products that can be classified as conjugate acids and bases.
Conjugate Acid: a base that gains a proton
Conjugate Base: an acid that loses a proton (gives it to a base)
For example, in this equation
HCL + H2PO4-=Cl- + H3PO4
HCL is the acid and H2PO4 is the base on the left side of the equation. On the right side of the equation, CL- is the conjugate base because it is derived from the HCL losing and H+ proton. The H3PO4 is the conjugate acid because it is derived from the H2PO4 gaining an H+ proton.
Conjugate Acid: a base that gains a proton
Conjugate Base: an acid that loses a proton (gives it to a base)
For example, in this equation
HCL + H2PO4-=Cl- + H3PO4
HCL is the acid and H2PO4 is the base on the left side of the equation. On the right side of the equation, CL- is the conjugate base because it is derived from the HCL losing and H+ proton. The H3PO4 is the conjugate acid because it is derived from the H2PO4 gaining an H+ proton.
Alkali
An alkali is a base that is soluble in water. Besides alkalies, bases are insoluble in water.