Neutralization of Acids and Bases
A neutralization reaction is a reaction in which an acid and a base combine in such a way that they neutralize each other and create salt and water.
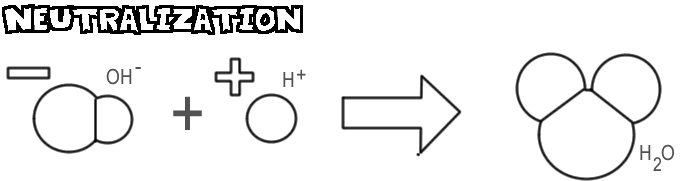
This reaction occurs when the moles (amount of chemical substance) of the acid and the moles of the base are equal. This makes the H+ from the acid and the OH- from the base combine and become water (see picture below), which cancels out the acidic and basic properties of the two substances.
This reaction occurs when the moles (amount of chemical substance) of the acid and the moles of the base are equal. This makes the H+ from the acid and the OH- from the base combine and become water (see picture below), which cancels out the acidic and basic properties of the two substances.
The entire neutralization reaction can be represented with the following equation:
Acid + Base --> Salt + Water
The salt produced depends on the acid involved in the titration, which you can learn more about on the making salts page
For example, a reaction of hydrochloric acid and sodium hydroxide in the right amounts so that they neutralize each other can be shown through the following equation:
NaOH + HCL--> NaCL + H2O
Base Acid Salt Water
Acid + Base --> Salt + Water
The salt produced depends on the acid involved in the titration, which you can learn more about on the making salts page
For example, a reaction of hydrochloric acid and sodium hydroxide in the right amounts so that they neutralize each other can be shown through the following equation:
NaOH + HCL--> NaCL + H2O
Base Acid Salt Water
Four Ways to Neutralize an Acid
Including combining it with a base, there are four ways to neutralize an acid. Here are the four ways and their corresponding equations
Here is a video showing a reaction between an acid and a metal
- Combine the acid with a metal, Acid + Metal --> Salt + H2
- Combine the acid with a carbonate, Acid + Carbonate --> Salt + H2O + CO2
- Combine the acid with an alkali, Acid + Alkali --> Salt + H2O
- Combine the acid with a base, Acid + Base --> Salt + H2O
Here is a video showing a reaction between an acid and a metal